Families are pleading with the federal health minister to approve a groundbreaking new drug that treats a rare and deadly type of childhood cancer.
Julia and Matthew Sammut’s son Harry is fighting a disease statistics say will likely take his life.
Neuroblastoma claims the lives of more children aged under five than any other cancer.
READ MORE: NDIS clients want provider who left them homeless and broke banned
There is a drug saving the lives of those with neuroblastoma overseas, DFMO, but it’s not available in Australia, meaning families are forced to fork out hundreds of thousands of dollars to travel to the US for treatment.
Harry was diagnosed with high-risk neuroblastoma – classed as a stage four cancer- at just eight years of age.
“Every time he got a test, it got worse,” Julia said.
“It was a tumour, it was in his bone, it was everywhere. The cancer was everywhere.”
For the next 18 months, he underwent six rounds of chemotherapy, surgery to remove his tumour, two bone marrow transplants, 12 rounds of radiation and immunotherapy.
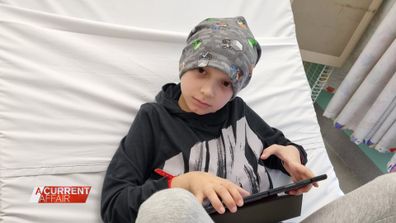
And despite all that, there’s still a 50 per cent chance that after treatment, Harry could relapse.
If he does, he has just a five per cent shot at surviving.
Harry’s parents scrimped and saved to send him to the US twice to be treated with DFMO.
It’s hoped it will save his life.
Neuroblastoma Australia chief executive Lucy Jones wants federal health minister Mark Butler to fund special access to the drug overseas before it’s approved in Australia, to stop sick kids from dying in the interim.

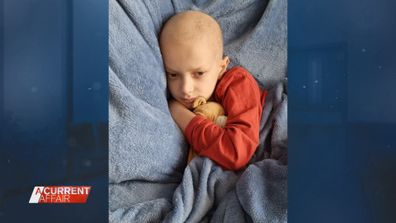
Jones knows the destruction of the disease, after losing her own daughter Sienna to neuroblastoma 14 years ago.
READ MORE: ‘The scheme is not wrecked’: NDIS minister responds to criticism claiming system is broken
“Children with neuroblastoma simply do not have the time for rare diseases, the regulatory approval process needs to be quicker,” Jones said.
Neuroblastoma Australia estimates it would cost the federal government between $5 million and $15 million dollars over two years to gain access to DFMO.
“$15 million is 0.1 per cent of the federal health budget,” Jones said.

“I think after the children have been through so much, to deny them this is just so cruel.”
With three more trips to America for treatment, Harry’s family is running out of cash and patience.
“Mark Butler, please help us get this drug into Australia to help all these beautiful Neuroblastoma children access the drug while they can to save their life,” Julia said.
In a statement, health minister Mark Butler said he wrote to pharmaceutical company Norgine urging them to apply to register DFMO in Australia and to consider compassionate access to Australian families in the meantime.
“Norgine have now lodged an application for DFMO with the TGA,” the statement said.
“They are now working with the TGA to achieve this as quickly as possible.”
But families like the Sammuts fear that process could take seven to 12 months – time kids with neuroblastoma don’t have.
A donation page has been set up to help the family in Harry’s Neuroblastoma journey.
To donate, head to the Rare Cancers Australia website.
FULL STATEMENT FROM HEALTH MINISTER MARK BUTLER
These stories are heartbreaking.
Neuroblastoma is a highly aggressive cancer often found in children with approximately 40 new child diagnoses each year in Australia.
Despite intensive treatment, the survival rate is only 50 per cent.
US Food and Drug Administration has recently approved a drug called difluoromethylornithine (DFMO) that has demonstrated excellent results in clinical trials.
Recently, the TGA granted DFMO Orphan Drug Designation which incentivises the sponsor to make the medicine available in Australia by exempting the company from a range of fees that normally apply to registration.
Pharmaceutical companies can also consider providing their product to these families through their compassionate access program.
I wrote to Norgine encouraging them to make an application to the TGA to register DFMO in Australia and to consider compassionate access to Australian families in the meantime and consider options to expand clinical trials in Australia.
Norgine replied recognising the severe unmet medical need in the treatment in Australia, and is committed to providing equity of access to all patients who are eligible for DFMO.
Norgine have now lodged an application for DFMO with the TGA. They are now working with the TGA to achieve this as quickly as possible.

