Weight-loss patients have expressed their disappointment at the government’s plan to enforce a blanket ban on replicas of the drug Ozempic later this year.
The ban will cut the supply to 20,000 Australians in October, amid growing patient safety concerns about the composition of the compounded drugs.
Queensland woman Bnette has had lap band surgery, the sleeve and a gastric bypass, but found semaglutide – the main ingredient in Ozempic – was the weight loss method that helped her lose 20 kilos.
READ MORE: Outback Wrangler Matt Wright faces off with best mate’s widow in court
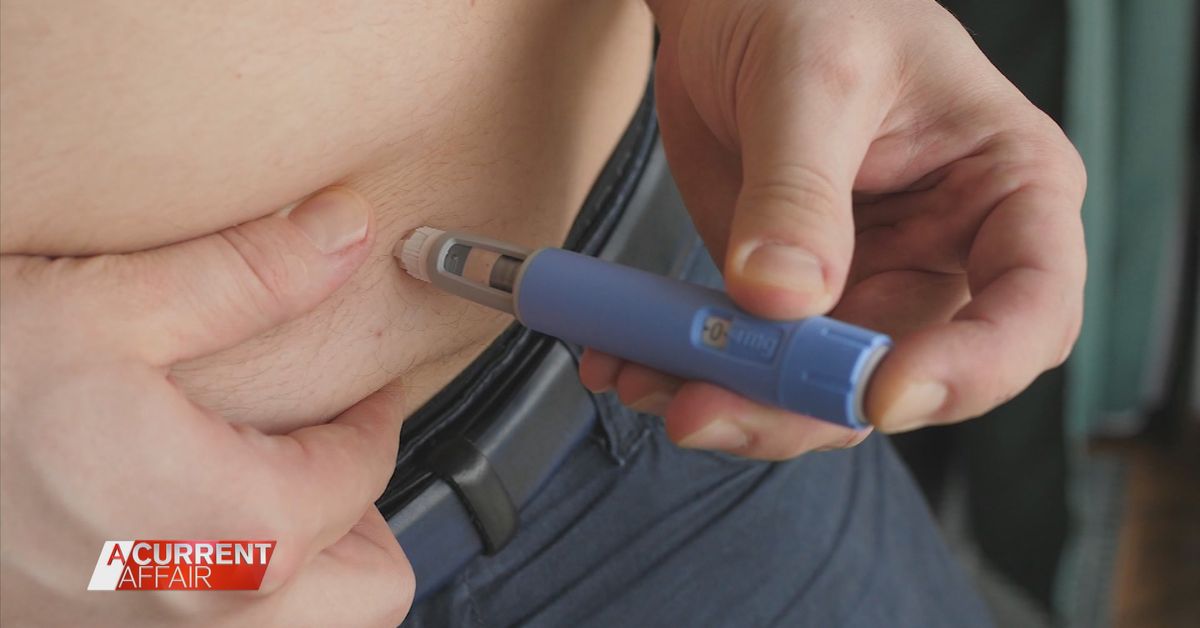
Ozempic is an approved medication made to improve blood sugar levels for type 2 diabetics.
But the once-a-week injection can also be an appetite suppressant, meaning it can also help some people lose weight.
“You actually eat only when you’re hungry. You don’t eat, you don’t snack during the day,” Bnette said of her own experience.
As word got around about the new weight-loss craze, it triggered a shortage of the drug, leaving people with diabetes desperately in need of their medication.
That’s why compounding chemists have legally been making replicas to meet demand.

Bnette said she was frightened that once the ban comes into effect in October, she won’t be able to maintain her current weight.
“It’s just changed my life so much. People around me say ‘you’re so happy, so bright and bubbly’,” she said.
“It’s just a game changer and for them to take that away… I just can’t fathom it.
“It’s not cheating and I’m not taking away from the diabetics who want medication.”
The problem for regulators is the commercial drive for the drug has meant cowboys have moved in, taking dangerous shortcuts.

“These are powerful new drugs that have a significant impact on the body and it’s simply not acceptable to have them compounded in a way that gets around all the safety regulations that have long existed in this country,” Health Minister Mark Butler said when he announced the ban.
Patients like Bnette need a doctor’s script in order for pharmacies to post the medicine in vials in the mail.
But Royal Australian College of General Practitioners (RACGP) associate professor Michael Clements said there was no assurance the correctly compounded medicines would be delivered to patients.
“They weren’t even compounding the original. They’re actually creating a new drug that wasn’t the original drug, it was a salt of it, calling it the same thing and then mailing it out to patients,’ Clements said.
The Therapeutic Goods Administration (TGA) recently raided a property in Sydney, where it’s alleged Ozempic replicas were being illegally manufactured in a kitchen.

“(There was) no oversight, no legislation, nobody checking on them, nobody testing the product afterwards or (testing) that it was safe,” Clements said.
“(They were) selling this and mailing it out to patients all in the names of profit.”
In good news for patients who use Ozempic replicas, Clements said a new TGA-approved drug could be just months away.
“We absolutely do have the products coming back,” Clements said.
“We absolutely expect new ones to come online and there’s even research into a tablet form of the same medication that we’re looking forward to.

“This is not a life-long ban. It’s not a lifetime ban. It’s a for-now ban.”
Whether or not Ozempic will help someone will depend on their personal circumstances.
Individuals should seek medical advice before considering taking any medication.
FULL THERAPEUTIC GOODS ADMINISTRATION (TGA) STATEMENT
The changes to the regulation of compounding glucagon like peptide-1 receptor agonists were based on advice from the Therapeutic Goods Administration (TGA), following a consultation process.
This is not about some pharmacies and poor production, there are genuine public safety concerns with the commercial-scale manufacturing of unregulated products.
The TGA evaluates manufactured medicines for safety, quality and efficacy. This includes rigorous “post-market” monitoring, including for adverse events experienced by patients.
These regulatory controls are crucial to provide the community and health professionals with confidence of the safety of medicines they access or prescribe.
However, compounded medicines are not regulated in the same way that large-scale manufactured prescription medicines are.
Compounded medicines are not evaluated for quality, safety or efficacy by the TGA.
Ingredients, manufacturing and the final product are not subject to the same requirements as TGA-approved medicines. Compounded medicines can have a different strengths and different ingredients from similar medicines approved by the TGA.
Additionally, compounded medicine are not subject to the same requirements for reporting adverse events, meaning we don’t get visibility of the extent of safety issues once they are being taken.
The large-scale manufacturing of medicines through compounding means that these products are being provided in large quantities, without the regulation and evaluation that such volumes should undergo.
Compounded semaglutide-like products supplied through compounding pharmacies are not identical or equivalent to the TGA approved products (such as Ozempic® or Mounjaro).
Pharmacy compounding of all applicable medicines is intended only for small quantities, at the request of a medical practitioner, for an individual patient with a prescription.
It is intended to meet the clinical needs of a particular patient, where a commercially manufactured medicine is either not suitable or not clinically appropriate.
The TGA is concerned that there are now over 20,000 patients who are being prescribed compounded GLP-1 RA copies, all outside our rigorous evaluation process.
The TGA has a role to evaluate and facilitate access to appropriate treatments.
However, when the risks outweigh the benefit of access, it recommends the appropriate regulatory actions to be taken.
The TGA will work with key medical, pharmacy and consumer stakeholders, and other health system regulators, to provide guidance in navigating the impact of these changes.
FULL PHARMACY GUILD OF AUSTRALIA STATEMENT
The Pharmacy Guild of Australia (Guild) always supports the safe and quality use of medicines, including pharmacists compounding medicines professionally and in accordance with Australian law.
Under current legislation, pharmacists must meet strict regulatory and professional requirements when compounding a medicine.
This includes:
– Compounding only for individual patients according to a prescription.
– Prohibition of batch compounding in anticipation of a prescription.
– Requirements for the compounding of sterile injectable medicines.
– Providing prescribers and patients with relevant information about the appropriateness and safety of the supply and compounding of a medicine.
Removing products from the pharmacist compounding exemption is not a substitute for enforcement of existing rules and legislation.
Given the ongoing shortage of commercial semaglutide worldwide, compounded products may be the only solution for some patients lacking alternate options.
The Guild supports the regulator working with community pharmacy to:
– Educate and inform community pharmacy on compliant compounding practice
– Provide prescribers and pharmacists with evidence-based information on compounding practice specific to semaglutide
– Actively monitor and prosecute non-compliance.
Legislative changes that remove the current exemption allowing community pharmacists to compound GLP-1 RAs set a concerning precedent, and fail to address the problem at hand.