CHAPEL HILL, N.C. – Angelman syndrome is a rare genetic disorder caused by mutations in the maternally-inherited UBE3A gene and characterized by poor muscle control, limited speech, epilepsy, and intellectual disabilities. Though there isn’t a cure for the condition, new research at the UNC School of Medicine is setting the stage for one.
Ben Philpot, PhD, the Kenan Distinguished Professor of Cell Biology and Physiology at the UNC School of Medicine and associate director of the UNC Neuroscience Center, and his lab have identified a small molecule that could be safe, non-invasively delivered, and capable of “turning on” the dormant paternally-inherited UBE3A gene copy brain-wide, which would lead to proper protein and cell function, amounting to a kind of gene therapy for individuals with Angelman syndrome.
“This compound we identified has shown to have excellent uptake in the developing brains of animal models,” said Philpot, who is a leading expert on Angelman syndrome and a member of UNC Lineberger Comprehensive Cancer Center. “We still have a lot of work to do before we could start a clinical trial, but this small molecule provides an excellent starting point for developing a safe and effective treatment for Angelman syndrome.”
But these results, which were published in Nature Communications, mark a major milestone in the field, according to Mark Zylka, PhD, the W.R. Kenan Jr. Distinguished Professor of Cell Biology and Physiology at the UNC School of Medicine and Director of the UNC Neuroscience Center. No other small molecule compound has yet to show such promise for Angelman, he added.

Ben Philpot, PhD
Unlike other single-gene disorders such as cystic fibrosis and sickle-cell anemia, Angelman syndrome has a unique genetic profile. Researchers have found that children with the conditions are missing the maternally-inherited copy of the UBE3A gene, while the paternally-inherited copy of the UBE3A gene remains dormant in neurons, as it does in neurotypical individuals. Typically, UBE3A helps regulate the levels of important proteins; missing a working copy leads to severe disruptions in brain development.
For reasons that aren’t fully clear, the paternal copy of UBE3A is normally “turned off” in neurons throughout the entire brain. Thus, when the maternal copy of the UBE3A gene is mutated, this leads to a loss of UBE3A protein in the brain. Philpot and other researchers have theorized that turning on the paternal copy of UBE3A could help treat the condition.
Hanna Vihma, PhD, a postdoctoral research fellow in the Philpot lab and first author on the study, and colleagues screened more than 2,800 small molecules from a Pfizer chemogenetic library to determine if one could potently turn on paternal UBE3A in mouse models with Angelman syndrome.
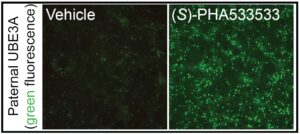
Researchers genetically modified mouse neural cells with a fluorescent protein that glows when the paternal UBE3A gene is turned on. After treating the neurons with more than 2,800 small molecules for 72 hours, researchers compared their thousands of treated cells against those treated with topotecan, a known small molecule that can turn on paternal UBE3A but lacked therapeutic value in animal models of the condition.
(S)-PHA533533, a compound that was previously developed as an anti-tumor agent, caused neurons to express a fluorescent glow that rivaled that induced by topotecan, meaning that its effect was potent enough to successfully turn on paternal UBE3A. Researchers were able to confirm the same results using induced pluripotent stem cells derived from humans with Angelman syndrome, indicating that this compound has clinical potential.

Hanna Vihma, PhD
Additionally, researchers observed that (S)-PHA533533 has excellent bioavailability in the developing brain, meaning it travels to its target with ease and sticks around. This is notable in that previous genetic therapies for Angelman syndrome have had more limited bioavailability.
“We previously showed that topotecan, a topoisomerase inhibitor, had very poor bioavailability in mouse models,” said Vihma. “We were able to show that (S)-PHA533533 had better uptake and that the same small molecule could be translated in human-derived neural cells, which is a huge finding. It means it, or a similar compound, has true potential as a treatment for children.”
Although (S)-PHA533533 shows promise, researchers are still working to identify the precise target inside cells that causes the desired effects of the drug. Philpot and colleagues also need to conduct further studies to refine the medicinal chemistry of the drug to ensure that the compound – or another version of it – is safe and effective for future use in the clinical setting.
“This is unlikely to be the exact compound we would take forward to the clinic,” said Philpot. Along with medicinal chemists in the lab of Jeff Aubé, PhD, the Philpot lab is working to identify similar molecules with improved drug properties and safety profiles. “However, this gives us a compound that we can work with to create an even better compound that could be moved forward to the clinic.”
This work was supported by the Angelman Syndrome Foundation, the National Institute of Neurological Disorders and Stroke, the National Institute of Child Health and Human Development, the Simons Foundation, a sponsored research agreement between the University of North Carolina and Pfizer (to BDP), and Pinnacle Hill, LLC, a portfolio company of certain funds managed by Deerfield Management Company, L.P (to BDP).